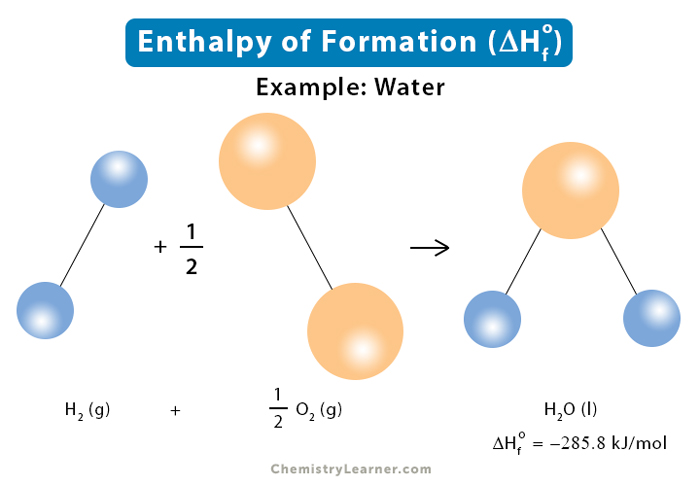
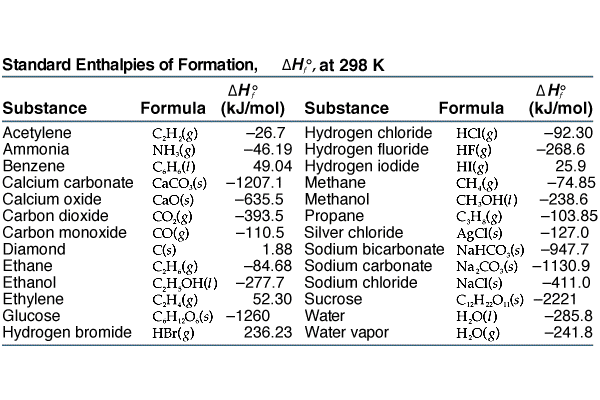
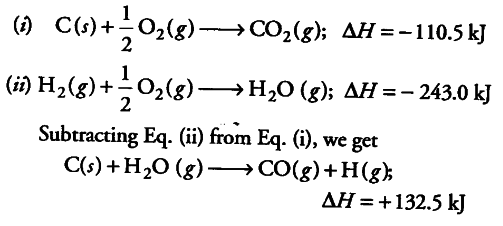Enthalpy of combustion of carbon to C${{O}_{2}}$ is -393.5 kj/mol. Calculate heat released upon formation of 35.2g of C${{O}_{2}}$ from carbon and ${{O}_{2}}$ gas? - CBSE Class 11 Chemistry - Learn CBSE
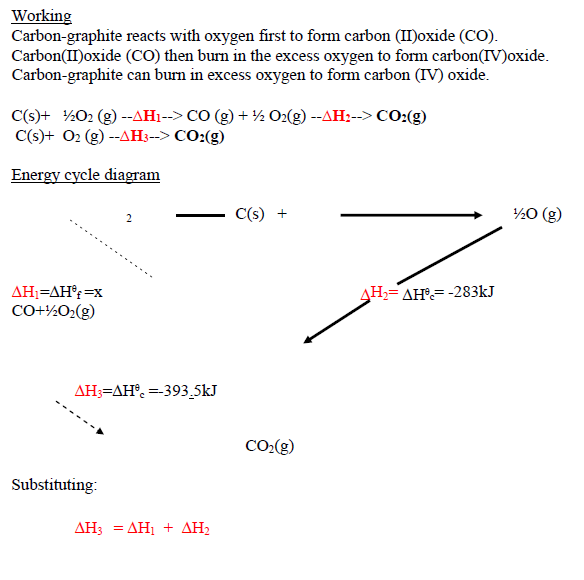
Calculate the molar enthalpy of formation of carbon(II)oxide (CO) given : ?H?c of carbon-graphite = -393.5kJmole<sup>-1</sup>, ?H?c of carbon(II)oxide (CO)= -283 kJmole<sup>-1</sup>

The enthalpy of formation for graphite as the function of the number of... | Download Scientific Diagram
![PDF] Prediction of Enthalpy of Formation in the Solid State (at 298.15 K) using Second-Order Group Contributions. Part 1. Carbon-Hydrogen and Carbon-Hydrogen-Oxygen Compounds | Semantic Scholar PDF] Prediction of Enthalpy of Formation in the Solid State (at 298.15 K) using Second-Order Group Contributions. Part 1. Carbon-Hydrogen and Carbon-Hydrogen-Oxygen Compounds | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c766ff3889423f02f154fa43603214c3fec1f15d/4-Table1-1.png)
PDF] Prediction of Enthalpy of Formation in the Solid State (at 298.15 K) using Second-Order Group Contributions. Part 1. Carbon-Hydrogen and Carbon-Hydrogen-Oxygen Compounds | Semantic Scholar

Why does the enthalpy of combustion for alkanes increase, e.g. from methane to propane, etc.? - Quora

Enthalpy of combustion of alcohols data trend graph explaining trend pattern determining delta H combustion comparison with ethers equations advanced A level organic chemistry revision notes doc brown