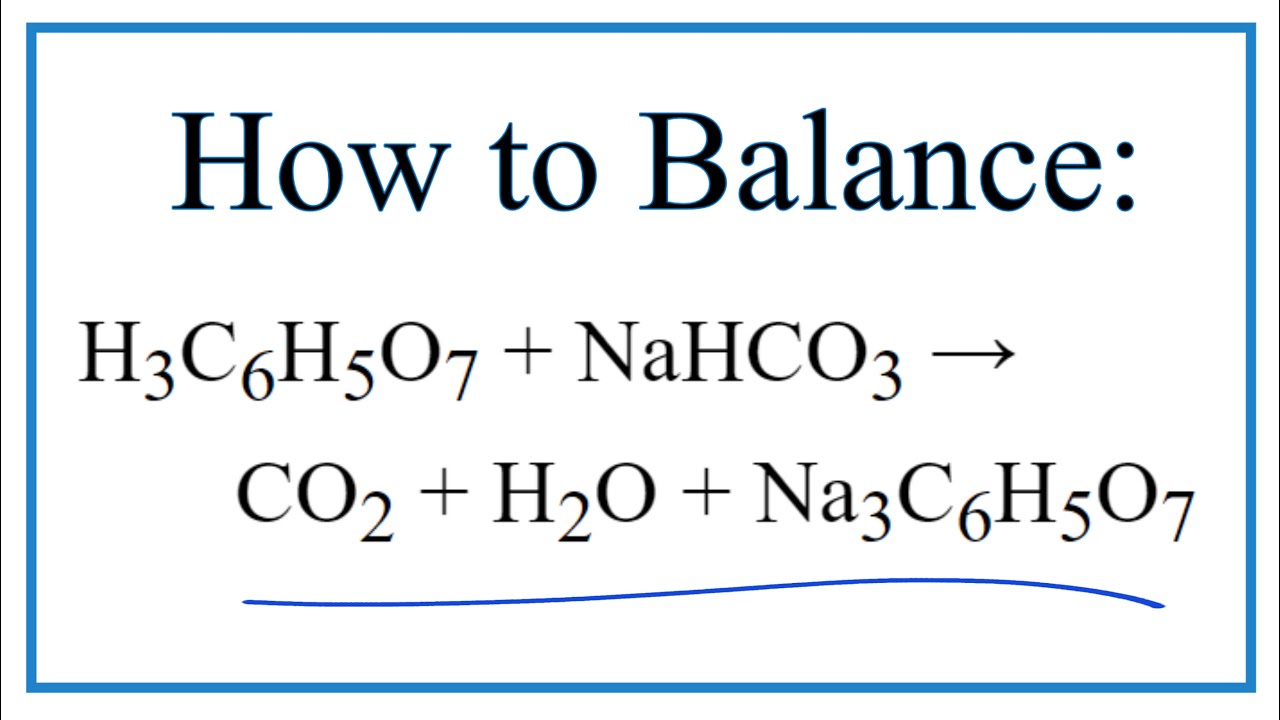
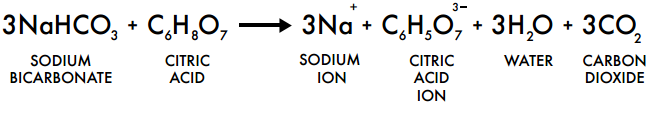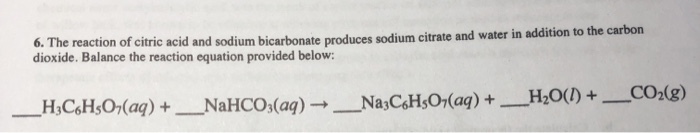A pneumatic power source using a sodium bicarbonate and citric acid reaction with pressure booster for use in mobile devices | Semantic Scholar
Exogenous citrate can react with carbonic acid to form citric acid and... | Download Scientific Diagram
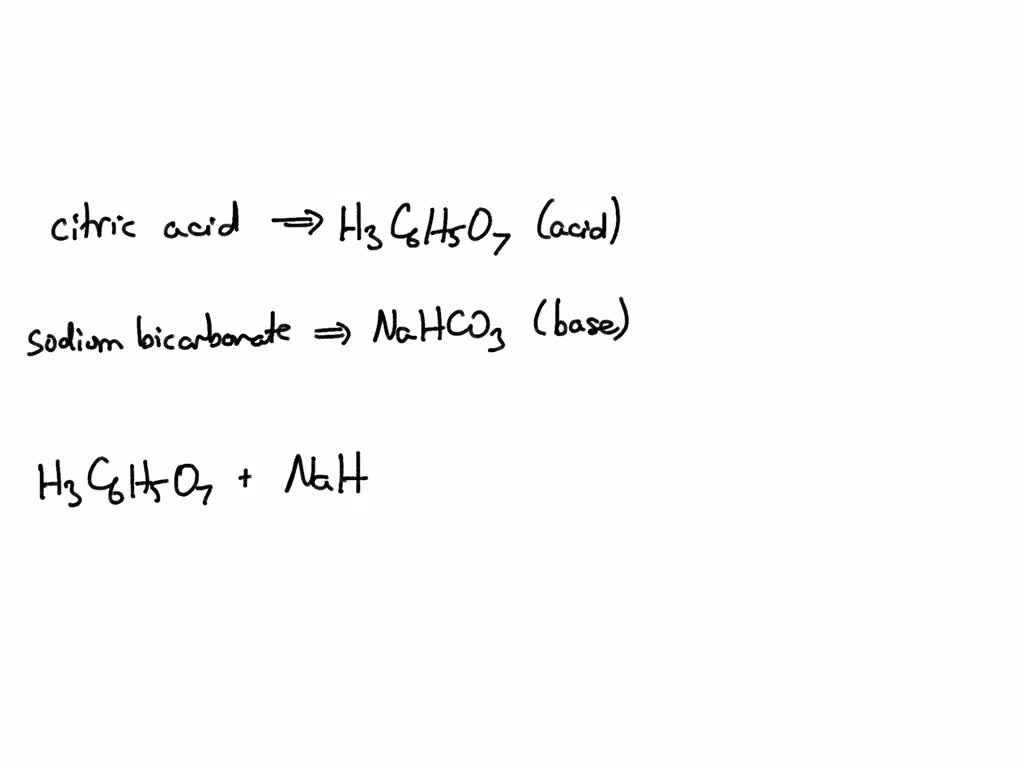
SOLVED: Write an equation for the neutralization of lemon juice (citric acid) with baking soda (sodium hydrogen carbonate or sodium bicarbonate). You equation should show a proton transfer to form carbonic acid

A pneumatic power source using a sodium bicarbonate and citric acid reaction with pressure booster for use in mobile devices | Semantic Scholar