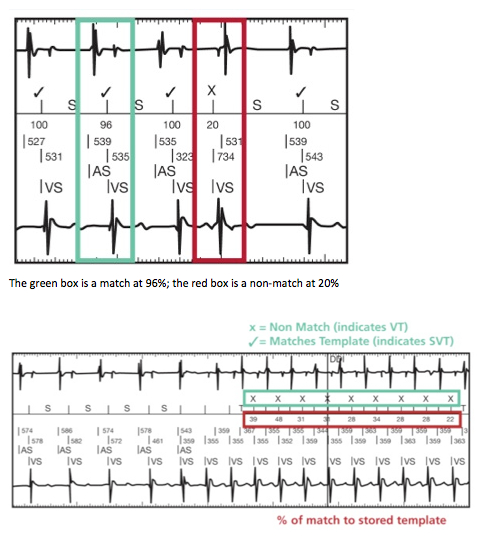
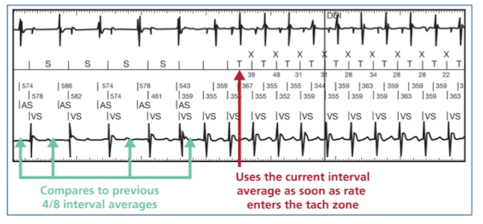
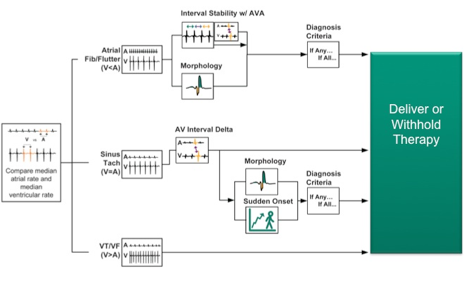
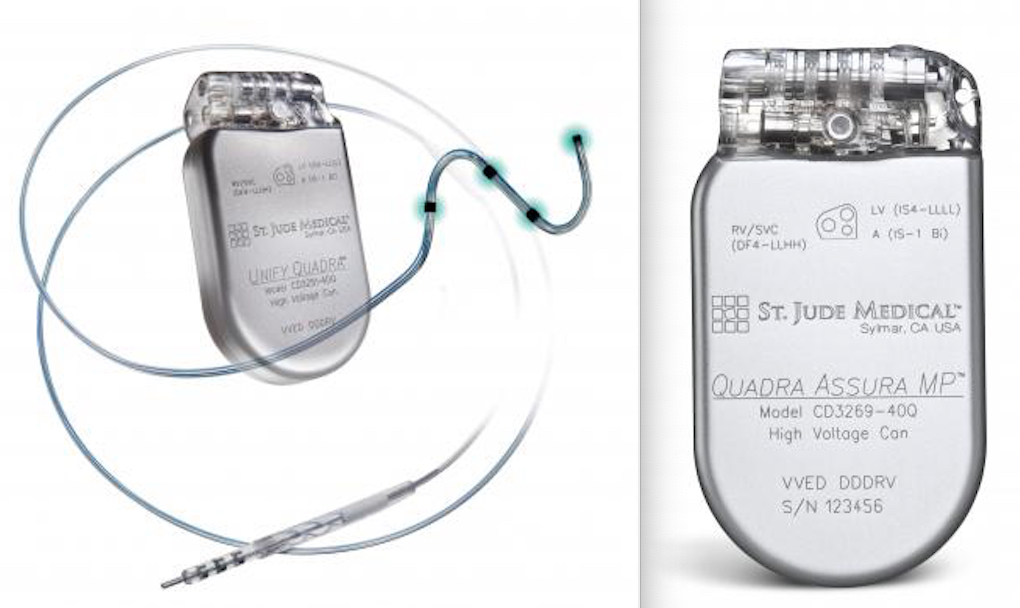
CD2357-40Q, St. Jude Medical, Implantable Cardioverter Defibrillator, Fortify Assura DR 40, Dual-chamber ICD with RF telemetry, Connector DF4-LLHH/IS-1

Micromachines | Free Full-Text | The Miniaturization of Cardiac Implantable Electronic Devices: Advances in Diagnostic and Therapeutic Modalities

JCM | Free Full-Text | Perioperative Management of Patients with Cardiac Implantable Electronic Devices and Utility of Magnet Application

PDF) Premature Battery Depletion in St. Jude Fortify® and Unify® Cardiac Implanted Electronic Devices

Recurrent implantable cardioverter-defibrillator shocks due to automatic deactivation of a right ventricular lead noise discrimination algorithm - ScienceDirect

CD1411-36C, St. Jude Medical, Implantable Cardioverter Defibrillator, Ellipse VR 36, Single-chamber ICD with RF telemetry, Parylene coating , Connector DF-1/IS-1